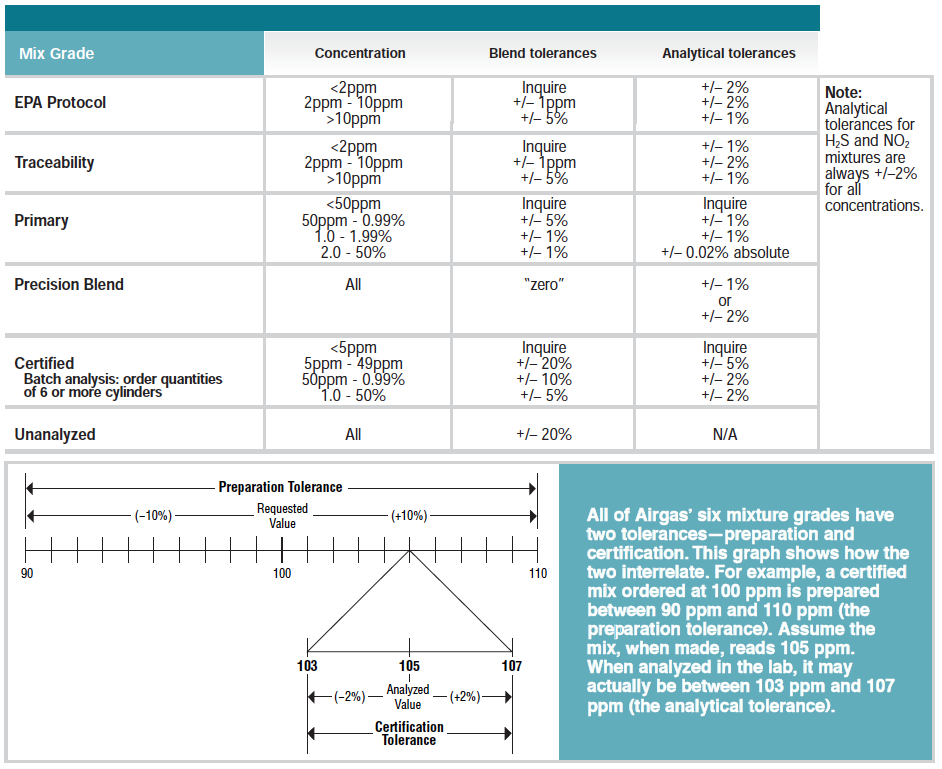
Our Gases
Rare and Specialty Gas
At MedOx we carry a full line of rare and specialty gases filled at state of the art facilities to ensure precision and accuracy. Whether it’s an NIST traceable, calibration standard, EPA protocol standard, laser gas, pure hydrocarbon, portable calibration or rare gas, we have the capabilities to meet the most stringent blend tolerances.
Traceability in Calibration Gas Mixtures
MedOx offers three grades of calibration gas mixtures with established and defined traceability to NIST or to an equivalent national measurement institute. Each traceable mixture is accompanied by full documentation in the form of a Certificate of Analysis (COA), designed in compliance with customer, regulatory, and ISO 17025 guidelines.
Traceability is defined as “the property of the result of measurement or the value of a standard whereby it can be related to stated references, usually national or international standards, through an unbroken chain of comparisons all having stated uncertainties.” A requirement for ISO 9001: 2000 programs, ISO 17025 compliant programs, emissions monitoring and reportable environmental testing is that the instrument calibration process maintain traceability to a national primary reference material.
Calibration gas mixture compositional traceability is assured through 2 methods:
1) Analytical traceability using Reference Materials from a national measurement institute (usually NIST) to calibrate the measurement system through a rigorous process to determine the concentrations of mixture components of interest, or
2) Process traceability to the international unit of mass (Kg) through comprehensive manufacturing and quality programs, using high precision, high sensitivity weighing systems for component additions. The resultant mixtures are analyzed versus Laboratory Primary Standards of known composition and uncertainty.
Typically, the process-based traceability is used when Reference Materials are unavailable from NIST or other national measurement institutes for the component(s) or concentration(s) of interest. Blends produced gravimetrically, using scales extensively calibrated with NIST certified weights, are considered traceable and have known uncertainty in their composition.
The majority of traceable products supplied by MedOx are certified to an overall analytical or process uncertainty not to exceed +/-1%.